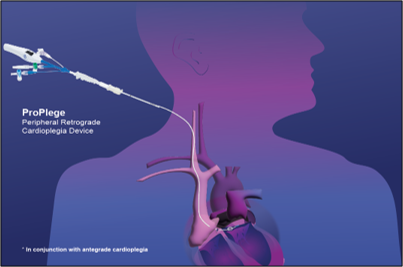
ProPlege
peripheral retrograde
cardioplegia device
Consistent delivery.
Global protection*.
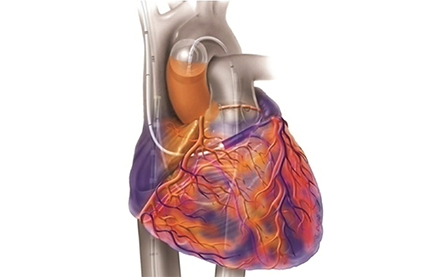
Adding ProPlege retrograde cardioplegia to antegrade delivery offers global myocardial protection without disruption, expanding minimally invasive cardiac surgery to more patients.
* In conjunction with antegrade cardioplegia
Benefits of the ProPlege device
Global protection*,
fewer complications
Paired with antegrade cardioplegia, ProPlege can
- More fully protect the heart1-4
- Help reduce risks of complications, such as cardiac and neurological events1-4
- Help patients come off bypass effectively for optimal hemodynamic return5,6
* In conjunction with antegrade cardioplegia
Maintain your
momentum
Streamline your procedures with reliable, uninterrupted performance
- Avoid redose disruption6
- Eliminate retractor repositioning1
- Avoid complex, inconsistent workarounds for each patient6,7
Designed for
minimally-invasive surgery
Enables minimally invasive on-pump cardiac procedures
- For more patients in a safer and more reliable manner than direct cannulation7,8
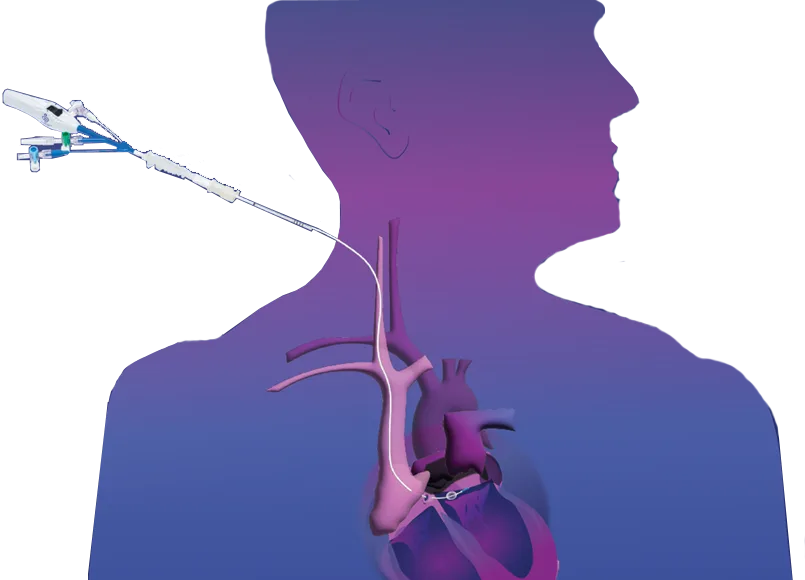
- Delivers retrograde cardioplegia through a peripheral approach outside the operating window
- With experience, anesthesiologists can reduce insertion time and optimize positioning6-10
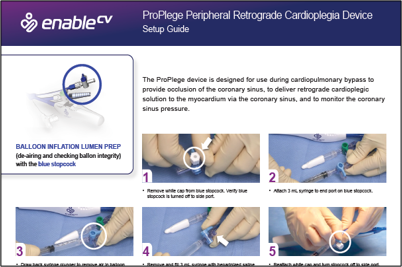
Device description & specifications
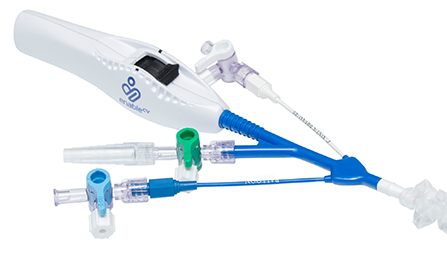
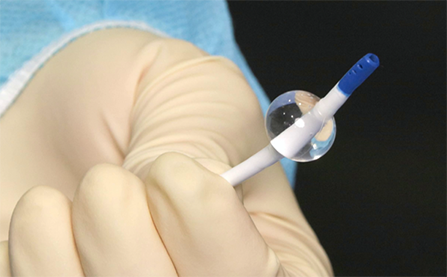
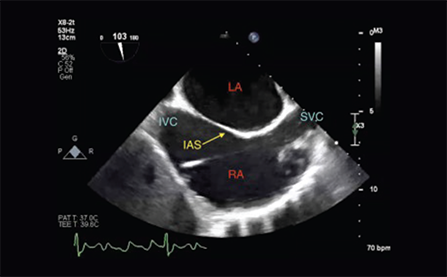
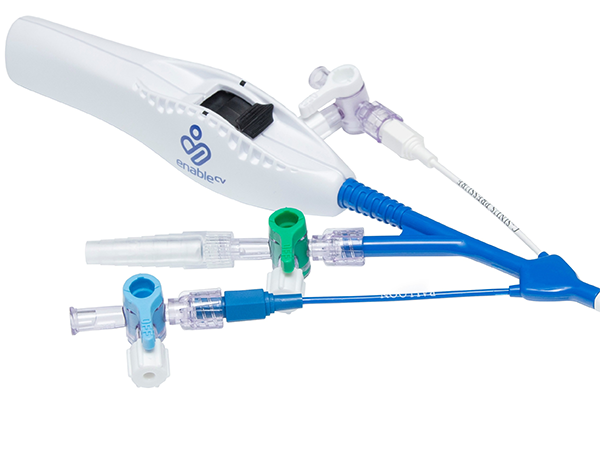
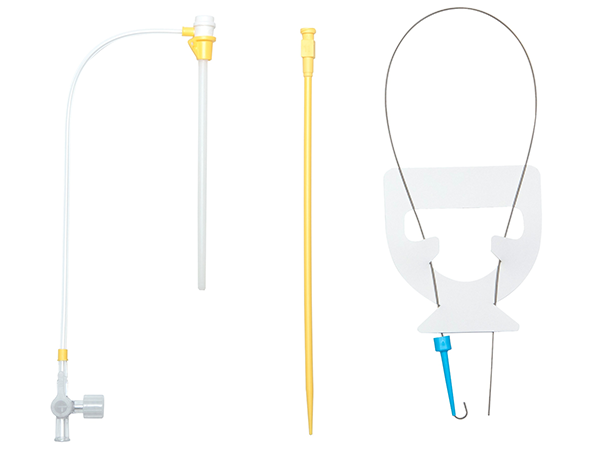
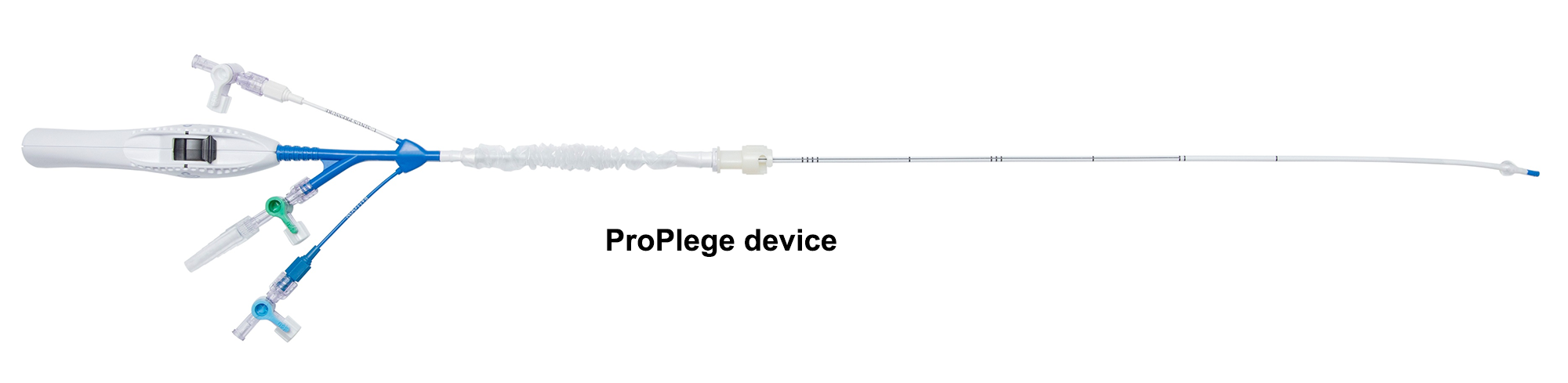
The ProPlege peripheral retrograde cardioplegia device (ProPlege device) is a 9 Fr (3.1 mm), 59 cm long, triple-lumen, articulating device with an elastomeric balloon near its tip for occluding the coronary sinus for retrograde perfusion of the coronary circulation.
The ProPlege device is indicated for:
- occlusion of the coronary sinus
- delivery of retrograde cardioplegia solution
- monitoring of coronary sinus pressure during cardiopulmonary bypass
Models & specifications
- Model no. PR9
- Catheter size – 9 Fr (3.1)
- Effective length – 23.2 in (59 cm)
- Overall length – 32.3 in (82 cm)
- The ProPlege device is intended for use ONLY
with a Cordis® AVANTI®+ 11 Fr introducer sheath- The Cordis® AVANTI®+ 11 Fr introducer sheath is sold separately by its manufacturer and is not included with the ProPlege device
- The Cordis® AVANTI®+ 11 Fr introducer sheath is sold separately by its manufacturer and is not included with the ProPlege device
ProPlege device prep video
Click the video for a demonstration on how to properly prepare the ProPlege peripheral retrograde cardioplegia catheter.
ProPlege device VR simulation
Click the video to experience using the ProPlege catheter in virtual reality.
References
1. Miller GS, et al. J Cardiothor Vasc Anes. 2010;24:743-5. 2. Khan AH, et al. J Cardiovasc Dis. 2015;13:10-4. 3. Loop FD, et al. J Thorac Cardiovasc Surg. 1992;104:608-18. 4. Dagenais F, et al. Ann Thorac Surg. 1999;68:1681-5. 5. Cernaianu AC, et al. Tex Heart Inst J. 1996;23:9-14. 6. Labriola C, et al. J Cardiothor Vasc Anes. 2017;31:1203-9. 7. Labriola C, et al. J Cardiothor Vasc Anes. 2015;29;598-604. 8. Lebon JS, et al. J Cardiothor Vasc Anes. 2019;33:1197-1204. 9. Ward AF, et al. J Thorac Cardiovasc Surg. 2014;148:2769-72. 10. Samarani G, et al. Am J Resp Crit Care Med. 2018;197:MeetingAbstracts.
Stay Connected
Please fill out this form to receive the latest information and updates from enableCV.